According to an international research led by Professor Toru Takumi (Senior Visiting Scientist, RIKEN Center for Biosystems Dynamics Research) and researcher Chia-wen Lin at Kobe University Graduate School of Medicine, the “debut ofautism idiopathic is triggered by epigenetic abnormalities in hematopoietic cells during fetal development, which causes immune dysregulation in the brain and gut.
The research findings highlighted that in autism there are immune abnormalities that can be observed in the brain and intestines. Further classification of the pathophysiology of autism could lead to the creation of new treatment strategies for autism and other neurodevelopmental disorders.
The results of the study have been published in the scientific journal Molecular Psychiatry.
Onset of autism: does it occur in the fetal stage?
Autism spectrum disorder is a developmental neurological condition that still needs to be devoted to studies and energy despite the rapidly increasing number of patients. Immune changes, now considered the cause of many diseases, also play an important role in the onset of autism.
Brain inflammation and peripheral immune system disorders have frequently been found in patients diagnosed with autism. Furthermore, the immune abnormalities are accompanied by abnormalities in the gut microbiota, which is also thought to be involved in the pathogenesis of the disease via the brain-gut axis. However, the essential mechanisms underlying these immune abnormalities remain to be elucidated.
Given the critical stages of development and the extensive involvement of the immune system in the onset of autism, the research team had an insight: a common etiology underlies widespread immune dysregulation and originates in several types of progenitor cells.
The analysis focused on the hematopoietic cells from which the immune cells derive, as well as the yolk sac (YS) and the aorta-gonad-mesonephros (AGM), which are involved in hematopoiesis during the fetal phase. These findings look for a common cause of inflammation in the brain and abnormalities in the peripheral immune system. In this study, BTBR mice were used as an idiopathic model for autism.
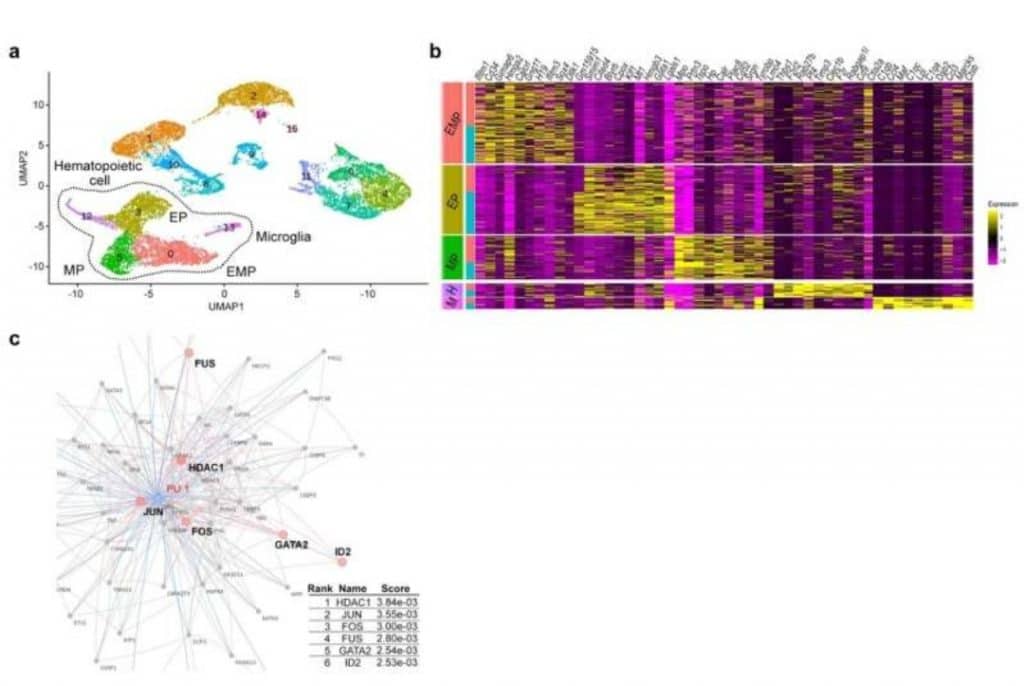
Single-cell RNA (sc-RNA seq) sequencing of BTBR mice traced the origin of immune abnormalities down to the embryonic stages of the yolk sac and aorta-gonad-mesonephro (AGM) and identified where macrophages differ ( microglia) and peripheral immune cells. Definitive hematopoiesis in single cell YS and AGM analysis successfully identified pathological mechanisms at the molecular level within rare progenitor cells in the early stages of development. Namely, the researchers recognized a common mechanism of transcriptional regulation through HDAC1, a histone deacetylase, underlying these pathologies.
The research team also showed that manipulating epigenetic dynamics during specific developmental stages can restore immune abnormalities in the brain and peripheral tissues. In essence, scientists have identified histone deacetylase HDAC1 as a common mechanism. Administration of this histone inhibitors (sodium butyrate or romidepsin) during the fetal phase in BTBR mice suppressed elevated inflammatory cytokines and activation of microglia.
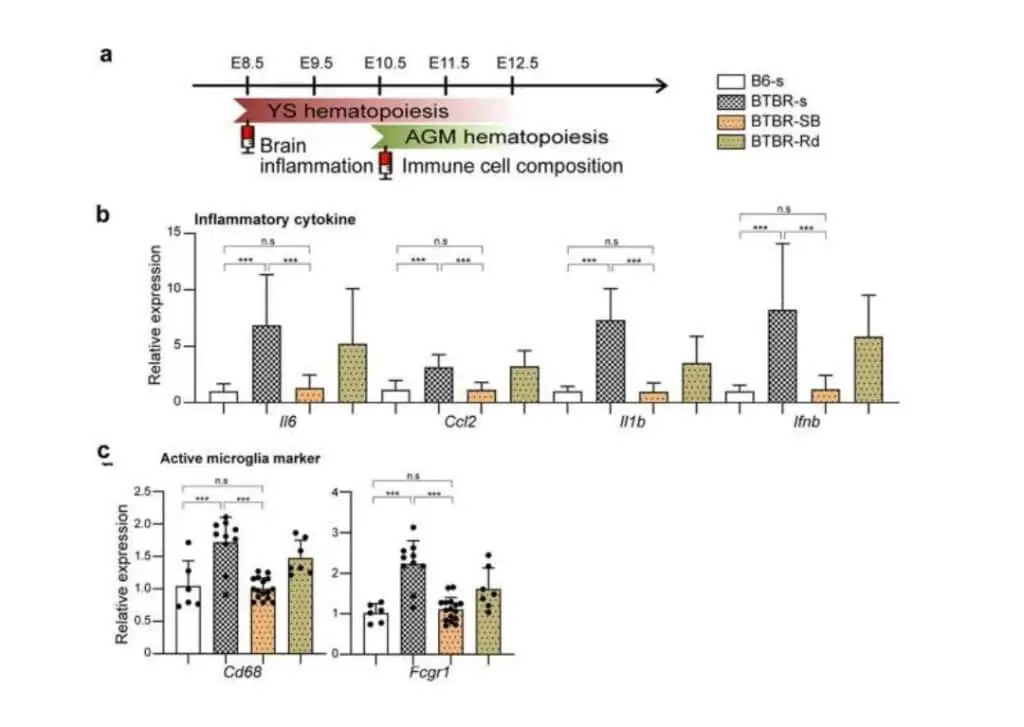
Not only that, the scientists showed that they demonstrated that dysregulated immunity can determine intestinal dysbiosis of specific profiles in ASD mice, which makes potential biomarkers of Treg and intestinal dysbiosis a means to classify the immuno-dysregulated ASD subtype. All this suggests that the abnormalities in the brain and peripheral organs (such as the gut) seen in autism are caused by epigenetic abnormalities in the lineage of hematopoietic stem cells, the ancestors of immune cells.
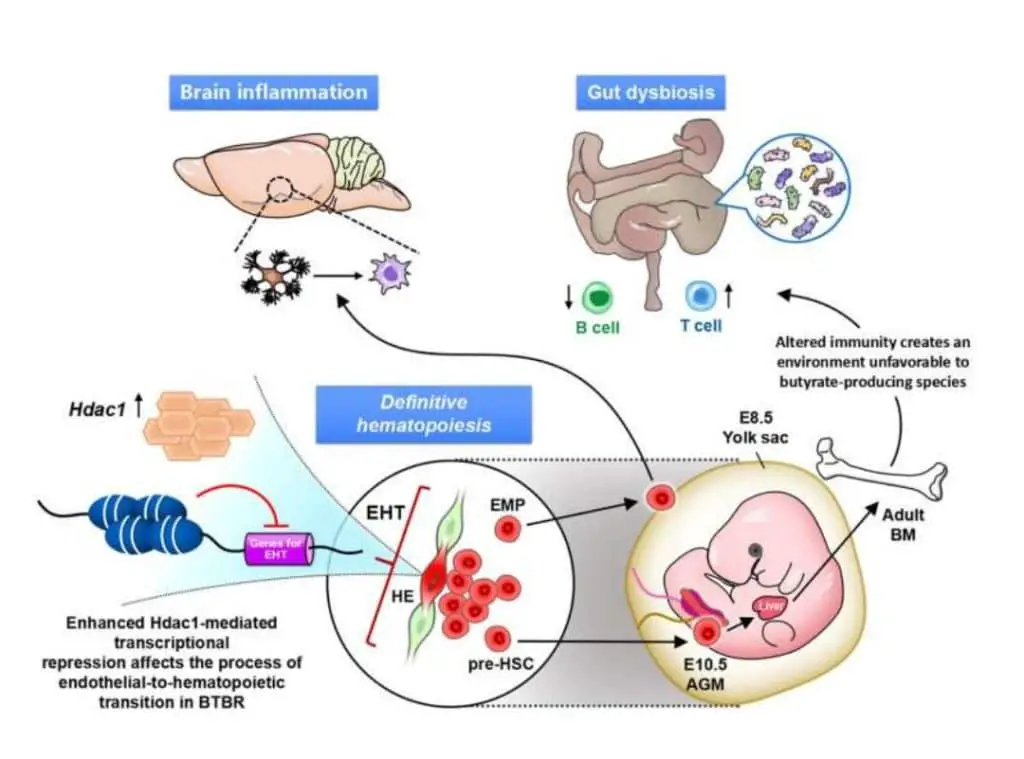
Evidence obtained during the research not only provides the missing piece to solving the long-standing puzzle of systemic immune dysregulation in autism, but also suggests the role of epigenetic disorder as a common etiology among different autism models of environmental risk factors.
#onset #autism #trigger #womb






