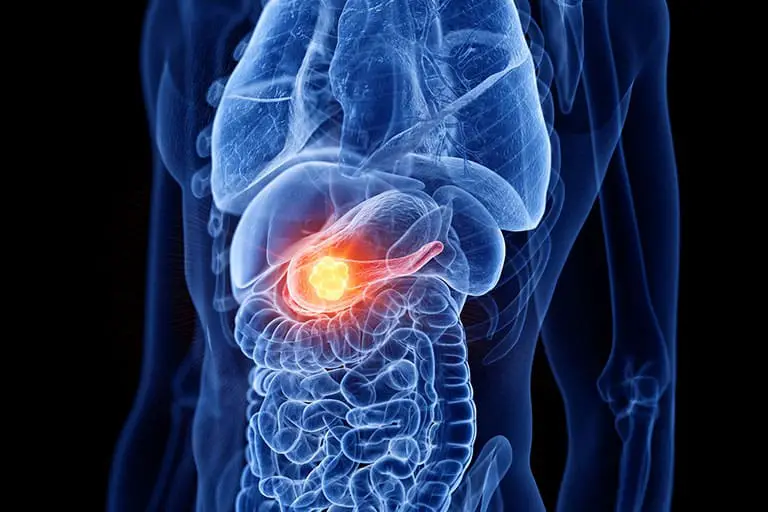
Researchers at Weill Cornell Medicine, along with an international team, used liver biopsies to identify cellular and molecular markers that could potentially predict if and when pancreatic cancer will spread to an individual’s liver or elsewhere, such as the lungs.
The study, published on Nature Medicine, suggests that information obtained from a liver biopsy – a small sample of tissue collected for laboratory analysis – when pancreatic cancer is diagnosed, can help guide doctors in tailoring treatment, such as liver-directed immunotherapies, before tumor cells have the ability to survive. possibility of metastasizing.
Only 10% of people with pancreatic cancer will survive more than two years after their initial diagnosis.
“If we could predict the timing and location of metastases, this could be a real game changer in the treatment of pancreatic cancer, particularly in patients at high risk of metastasis,” said study co-senior author David Lyden, MD, the Stavros S. Niarchos Professor of Pediatric Cardiology and professor of pediatrics and of developmental and cell biology at Weill Cornell Medicine.
In 2015, Dr. Lyden and his colleagues discovered that pancreatic cancer cells secrete factors that travel to distant organs, most often the liver, to create a premetastatic niche in which new tumors can form.
To find out how these alterations trigger their new location for cancer colonization, Dr. Lyden teamed up with lead author Dr. Linda Bojmar, an adjunct research assistant professor of molecular biology in pediatrics at Weill Cornell Medicine and an assistant professor of clinical and experimental medicine at Linköping University in Sweden.
Together with researchers at Memorial Sloan Kettering Cancer Center, including co-senior author Dr. William Jarnagin, co-first authors Drs. Constantinos Zambirinis and Jonathan Hernandez, and the hepatopancreatobiliary team, Dr. Lyden and his team obtained liver biopsies from 49 individuals undergoing surgery for early-stage pancreatic cancer. They also collected liver biopsies from 19 people undergoing similar surgery for conditions unrelated to cancer, such as the removal of benign pancreatic cysts.
The researchers then conducted a series of molecular, cellular and metabolic analyzes on these samples to determine whether they could identify hallmarks that preceded, or potentially prevented, subsequent metastases in the patients. They found that the livers of recurrence-free survivors, who showed no signs of the cancer spreading after a follow-up period of at least three years, closely resembled the livers of people who had never had cancer.
At the other end of the spectrum were those who developed liver metastases within six months of diagnosis, a group of patients who have a poor prognosis and limited treatment options. Their livers were filled with so-called neutrophil extracellular traps (NETs), dense tangles of DNA and enzymes released by dying neutrophils, immune cells that are a first line of defense against infection. Because these NETs are strongly associated with future metastases and develop so early in the disease, radiological imaging may in the near future be able to detect them and identify patients at risk for this aggressive spread.
“These individuals could then receive a full course of chemotherapy or, if metastases are detected when only a few appear, perhaps the secondary tumors could be surgically removed,” said Dr. Lyden, who is also a member of the Sandra and Edward Meyer. Cancer Center and the Gale and Ira Drukier Institute for Children’s Health at Weill Cornell Medicine. Additionally, he and his colleagues are studying whether drugs that digest the DNA that forms NETs could prevent liver metastases.
The researchers identified two other categories of patients in the study: those who would later develop metastases to the liver and those who would have had the cancer spread to other sites, such as the lung. Patients whose tumors spread to organs other than the liver showed a strong immune response against the cancer: protective T cells and natural killer cells had infiltrated their livers, and many immunoregulatory genes had been activated. These individuals who are prone to developing metastases outside the liver may benefit from immunotherapy to boost their ongoing anti-tumor immune response.
On the other hand, those whose livers succumbed to subsequent metastases also accumulated immune cells, but the cells showed signs of metabolic exhaustion. “It’s as if the liver was trying to protect itself, but ultimately lost the battle,” said Dr. Bojmar.
The researchers plan to validate their findings in a larger cohort of pancreatic cancer patients and examine whether this approach might be useful with other newly diagnosed cancers.
“We hope to develop a tool to predict which colorectal cancer patients will develop liver metastases based on the cellular, molecular, and metabolic profiles of their liver biopsies,” said co-senior author Dr. Robert Schwartz, associate professor of medicine at Weill Cornell Medicine.
Authors from several institutions in the United States, Sweden, and Israel contributed to this study.
A research article published in Nature Cancer details new insights into the role of efferocytosis – the burying of dead cells – in pancreatic cancer that spreads to the liver.
Liver metastases occur in 40-50% of patients with pancreatic ductal adenosarcoma (PDAC), and there are currently no effective therapies to cure pancreatic cancer patients who have liver metastases.
Led by Professor Michael Schmid of the University of Liverpool and his colleagues, this study found that PDAC metastases have high levels of immunosuppressive macrophages, a type of white blood cell that promotes tumor growth.
The researchers found that blocking the efferocytosis pathway during early-stage metastasis prevented this immunosuppressive activity in macrophages, restoring T-cell activation and reducing metastatic tumor burden.
Lead author Professor Michael Schmid said: “In pancreatic cancer, malignant tumor cells often spread to the liver. Our data show that the generation of a supportive metastatic ‘niche’ in the liver is crucial for effective malignant tumor growth.” cells at the distant site.
“Our findings suggest that a particular type of immune cell orchestrates the formation of the metastatic niche by reprogramming other immune cells, thereby creating an immunosuppressed metastatic microenvironment, in which malignant cells are able to hide from an antitumor immune response. Targeting this particular type of innate immune cells or interfering with their immunosuppressive functions could serve as a promising therapeutic approach for patients with metastatic pancreatic cancer.”
First author Dr Yuliana Astuti added: “Using single-cell technologies, we discovered an underappreciated diversity of macrophages in pancreatic cancer liver metastases. We identified that metastasis-associated macrophages with opposing functions coexist in the liver, some displaying immunostimulatory and others immunosuppressive characteristics.”
“Interestingly, further temporal analysis revealed that liver metastases are accompanied by increased liver tissue cell death and that engulfment of dead cells acts as a key factor to reprogram macrophages towards an immunosuppressive phenotype. Our study provides proof of principle that tailored targeting of specific macrophages restores tumor immunity and inhibits PDAC metastasis.”
Key collaborator Professor Ainhoa Mielgo commented: “Pancreatic cancer is a very aggressive type of cancer that often spreads to the liver. We currently have no effective therapies to treat pancreatic cancer patients who have liver metastases. These findings are truly exciting because they reveal a remarkable mechanism by which pancreatic cancer cells spread and grow in the liver. Our hope and goal now is to translate these laboratory discoveries to the benefit of patients.”
“This study is the result of an extraordinary collaborative effort by scientists, medical oncologists, surgeons and patients who worked together to find better treatments for pancreatic cancer patients,” added Professor Schmid.
Led by the University of Liverpool, the study also involved researchers from the Cancer Research-UK Scotland Institute, the University of Glasgow and the University of Edinburgh.
#Pancreatic #cancer #cellular #markers #predict #metastasis













