During a research carried out by the scientists of the‘University of Linköping (LiU) and LinkoCare Life Sciences AB a bioengineered cornea based on pig skin collagen proteins. The implant restored the vision of 20 people with diseased corneas, most of whom were blind before receiving the implant.
The very encouraging results are a hope for those affected by corneal blindness And low vision providing a bioengineered implant as an alternative to human corneas donate, which are in short supply in countries where their need is greatest.
The results of the study have been published in the scientific journal Nature Biotechnology.
Bioengineered cornea: this is how it works
“The results show that it is possible to develop a biomaterial that meets all the criteria for use as human implants, which can be mass-produced and stored for up to two years and thus reach even more people with vision problems. This helps us solve the problem of a shortage of donated corneal tissue and access to other treatments for eye diseases, “he said. Neil Lagali, Professor at the Department of Biomedical and Clinical Sciences of the LiU, one of the researchers behind the development of the bioengineered cornea.
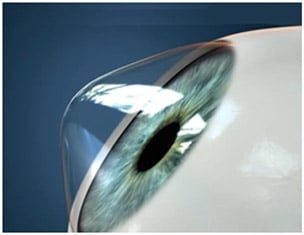
It is estimated that around 12.7 million people worldwide are affected by blindness due to their corneas, which is the outermost transparent layer of the eye, damaged or diseased. Their only way to recover their sight is to have a cornea transplanted from a human donor. But only one in 70 patients receives a cornea transplant. Additionally, most corneal transplant recipients live in low- and middle-income countries where access to treatment is very limited.
“The safety and effectiveness of bioengineered plants have been the focus of our work,” he explained Mehrdad Rafat, the researcher and entrepreneur behind the design and development of the bioengineered cornea. Rafat is Adjunct Associate Professor (Senior Lecturer) in the Biomedical Engineering Department of LiU e founder and CEO of LinkoCare Life Sciences ABwhich produces the bioengineered corneas used in the study.
“We have made significant efforts to ensure that our invention is widely available and affordable to everyone and not just the rich. That’s why this technology can be used in all parts of the world, ”added Rafat.
The cornea was primarily designed using protein collagen. To create an alternative to the human cornea, the researchers used collagen molecules derived from pig skin that were highly purified and produced under strict conditions for use in humans. The pork skin used is a by-product of the food industry, which makes it easy to access and cost-effective.
In the process of constructing the implant, the researchers stabilized the dissolved collagen molecules to form a sturdy, transparent material capable of resisting manipulation and implantation in the eye. Although donated corneas must be used within two weeks, the bioengineered cornea can be stored for up to two years before use.
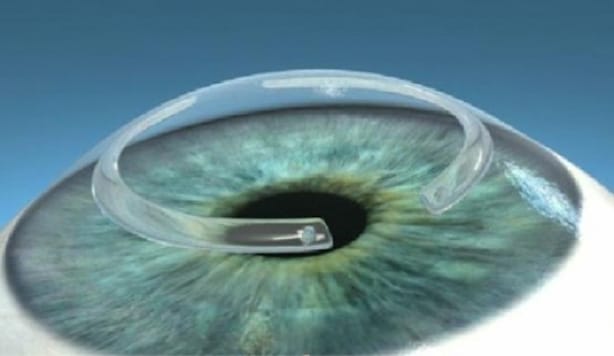
The team of scientists also designed a new, minimally invasive method for treating keratoconus disease, in which the cornea becomes so thin that it can lead to blindness. Today, the cornea of a patient with advanced stage keratoconus is surgically removed and replaced by a donated cornea, which is stitched into place using surgical sutures. This type of surgery is invasive and is only performed in larger university hospitals.
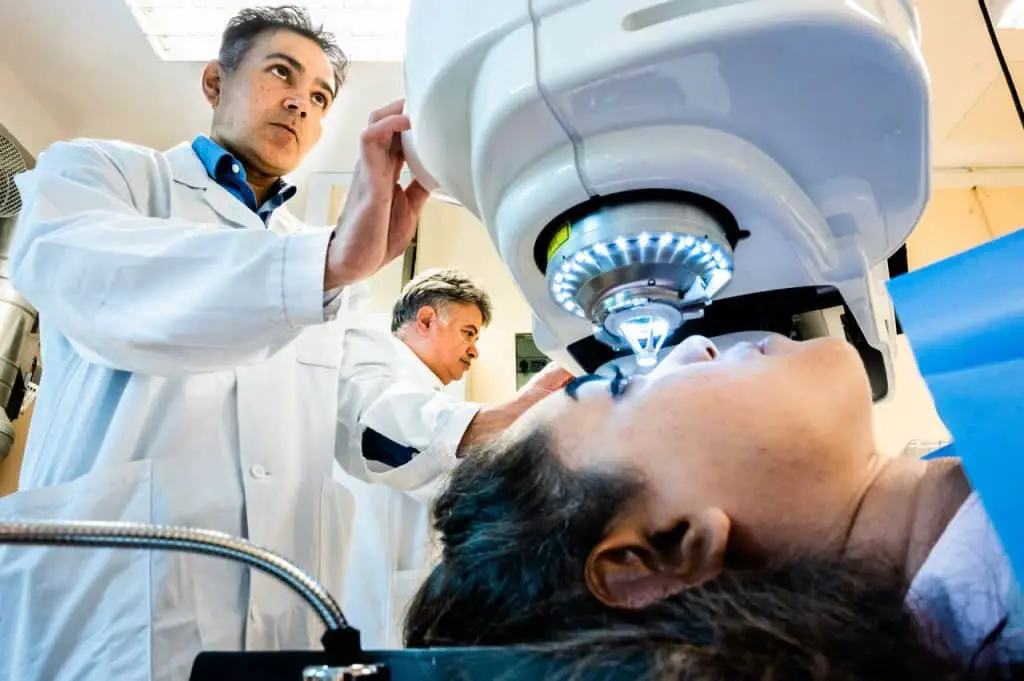
“A less invasive merodo could be used in more hospitals, thus helping more people. With our method, the surgeon does not need to remove the patient’s tissue. Instead, a small incision is made, through which the implant is inserted into the existing cornea, ”said Neil Lagali, who led the research team that developed this surgical method.
No stitches are required with this new surgical method. The incision in the cornea can be performed with high precision thanks to an advanced laser, but also, when necessary, by hand with simple surgical instruments. The method was first tested in pigs and proved to be simpler and potentially safer than a conventional cornea transplant.
The surgical method and bioengineered cornea have been used by surgeons in Iran and India, two countries where many people suffer from corneal blindness and low vision, but where there is a significant lack of donated corneas and treatment options. Twenty people who were blind or on the verge of losing their sight due to advanced keratoconus participated in the pilot clinical study and received the biomaterial implant.
The operations were uncomplicated and the tissue healed quickly. An eight-week treatment with immunosuppressive eye drops is then performed, which has been shown to be sufficient to prevent implant rejection. With conventional corneal transplants, anti-rejection therapy must be administered for several years. Patients were followed up for two years and no complications were noted during that time.
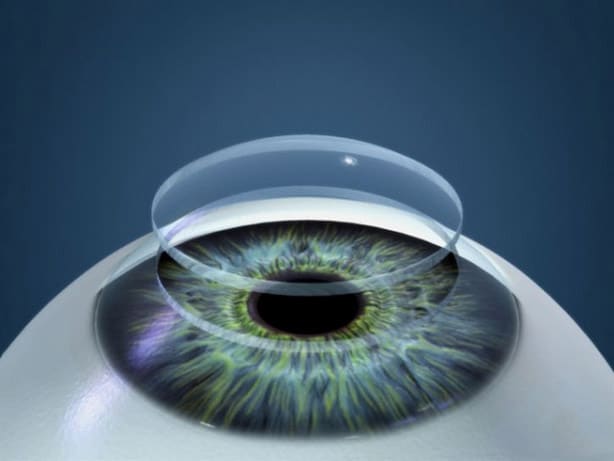
The main purpose of the pilot clinical study was to verify whether the bioengineered cornea was safe to use. However, the researchers were surprised at what happened with the implant. The thickness and curvature of the cornea were restored to normal. At the group level, participants’ vision improved as much as it would have been after a corneal transplant with donated tissue. Before the operation, 14 of the 20 participants were blind. After two years, none of them were blind anymore. Three of the Indian participants who were blind before the study and have perfect vision (20/20) after implantation of the bioengineered cornea.
A larger clinical trial followed by market approval by regulatory authorities is needed before bioengineered cornea can be used in healthcare. The researchers also want to investigate whether the technology can be used to treat more eye diseases and whether the implant can be tailored to the individual for even greater effectiveness.
#Bioengineered #cornea #returns #sight












