A team of neurologists, stem cell specialists and molecular biologists affiliated with several institutions in the United States and led by a team from the Stanford University School of Medicine, has found evidence that the root cause of disease of Alzheimer's could be the accumulation of fat in brain cells.
The study was published in the journal Nature.
New research on Alzheimer's disease
Previous research has suggested that Alzheimer's disease is caused by a buildup of beta-amyloid in plaques that grow between nerve cells. Other work has also implicated a protein called tau, which can accumulate in brain cells.
Therefore, most of the work done in developing methods to prevent, slow or stop the disease is based on reducing or eliminating these accumulations. But as researchers have discovered with this new effort, there may be something else at the root of the disease's development.
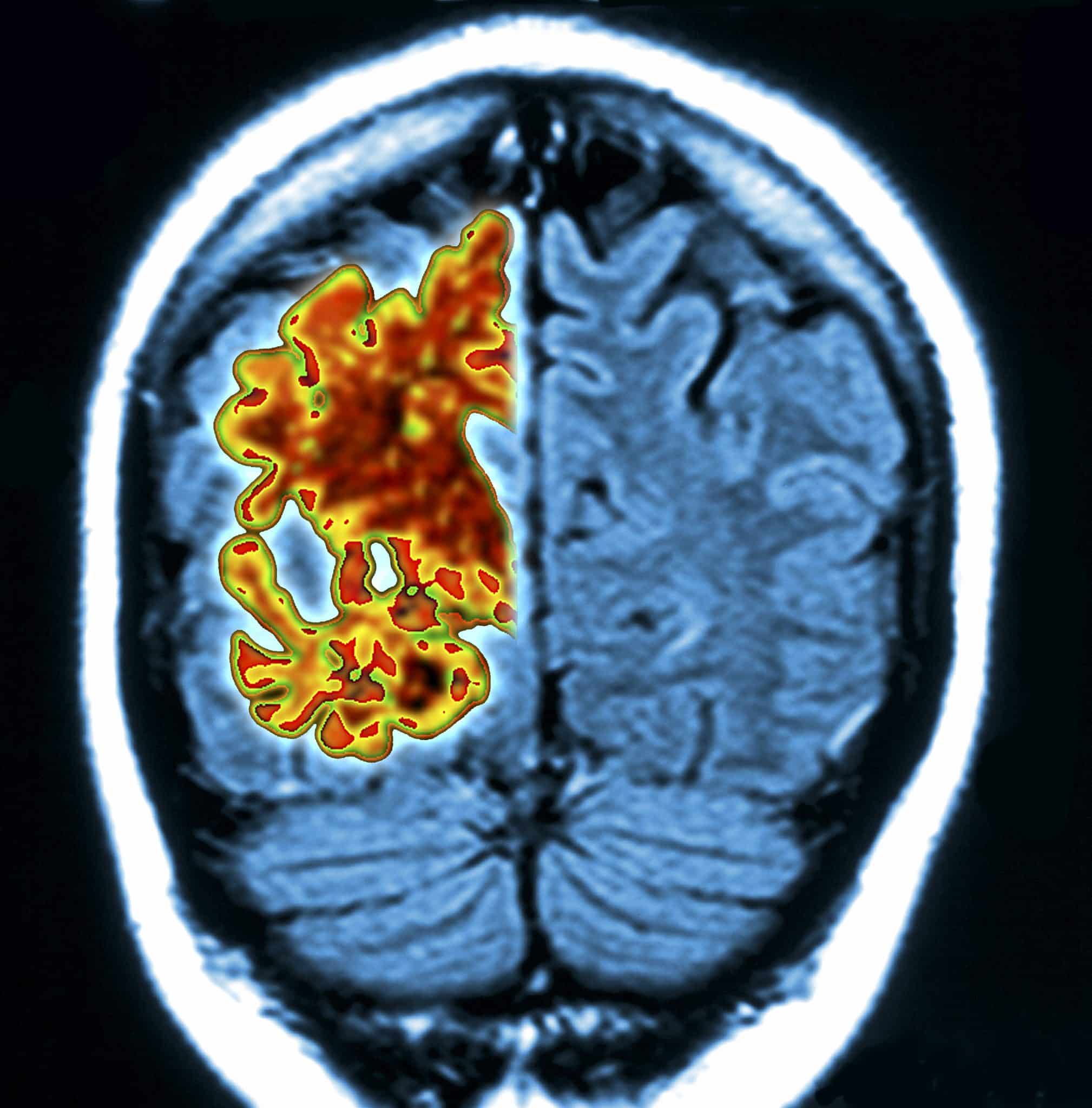
Back when Alzheimer's disease was first identified by Alois Alzheimer, he noticed that in addition to plaques and tau accumulation, there was also an accumulation of fat droplets in the brain cells.
Since then, little effort has been made to determine whether they could be the cause of the disease.
The research team then focused on the function of the APOE gene: previous research had shown that it encodes a protein involved in the transport of fat droplets into nerve cells.
Previous research has also shown that there are four variants of APOE, numbered 1 through 4, and that one of these, APOE4, carries the most fat into brain cells, while APOE2 carries less.
The team wondered whether APOE variants carried different risks for developing Alzheimer's disease. To find out, they conducted some experiments.
In the first experiment, the researchers used single-cell RNA sequencing to identify proteins within a test nerve cell.
They applied what they found to tissue samples collected from people who died of Alzheimer's who had double copies of APOE4 or APOE3.
They found that the brains of people with the APOE4 gene had more immune cells with a type of enzyme that increased the movement of fat droplets into brain cells.
In another experiment, they found that applying amyloid to the brain cells of people with the APOE4 or APOE3 variants caused the cells to accumulate more fat.
According to the researchers, the findings indicate that the buildup of amyloid in the brain triggers fat to be pushed into brain cells, leading to the disease.
Protein accumulation on fat droplets implicated in late-onset Alzheimer's disease
Sarah Cohen, Ph.D., a researcher at the UNC School of Medicine, and Ian Windham, a former Ph.D. student in the Cohen lab, have made a new discovery about apolipoprotein E (APOE), the largest genetic risk factor for late-onset Alzheimer's disease.
Older people who inherited a genetic variant called APOE4 from their parents are two to three times more likely to develop a late-onset neurodegenerative disease. If researchers can better understand how APOE4 affects brain cells, it could help them design effective therapies and target the mechanisms that cause increased disease risk.
Cohen and Windham conducted an exceptionally thorough, five-year study to better understand and visualize the relationship between APOE4, Alzheimer's disease, and fat molecules called lipids in the brain.
“We found that brain cells known as astrocytes are more vulnerable to damage and can even become dysfunctional when APOE4 surrounds their lipid storage centers,” said Cohen, assistant professor of cell biology and physiology and senior author of the article published in the Journal of Cell. Biology. “This mechanism could explain why APOE4 increases the risk of Alzheimer's at the cellular level.”
Sixty percent of the brain's dry mass is composed of lipids, which play important roles in the brain, such as storing cellular energy and forming myelin, which surrounds and insulates neurons. Lipids are found in specialized fat storage compartments known as lipid droplets within astrocytes.
As useful as they can be, lipids can also become toxic if the conditions are right. When excited or stressed, neurons release toxic lipids into the environment. Astrocytes are responsible for cleaning up floating toxic lipids and preventing them from accumulating in the brain.
If astrocytes were damaged or dysfunctional in some way, they would not be able to perform their cleaning functions. As a result, other brain cells, called microglia, also fail to clear beta-amyloid plaques in the brain, another driver of Alzheimer's disease.
APOE is produced by astrocytes. Just like a taxi or an Uber, the protein oversees the release and transport of lipids between cell types in the brain. Windham and Cohen wanted to see what exactly happens with the lipids in astrocytes. Windham led the initiative, creating a labeling and marking system that would allow them to see astrocyte guts in action under a microscope.
“Tagging APOE with a green fluorescent protein allowed us to see the different places APOE goes within living cells,” said Windham, now a postdoctoral researcher at Rockefeller University and first author of the article.
The team first fed the astrocytes with oleic acid, an omega-9 fatty acid produced naturally in the body. Using a microscope, the team observed the usual formation of lipid droplets. APOE4, surprisingly, approached the lipid droplets like a magnet and changed the shape and size of the droplets.
It has become apparent to researchers that APOE4 can escape secretion, lock within astrocytes, and migrate into lipid droplets within astrocytes.
Windham and Cohen hypothesize that the altered composition of lipid droplets could cause astrocyte dysfunction and affect the ability of microglia to clear amyloid beta.
More research is needed to know the details. Cohen hopes their findings will further highlight the role of lipid droplets in Alzheimer's disease and other neurodegenerative diseases.
“In Alois Alzheimer's first paper, he described three features of neurodegenerative disease: beta-amyloid plaques, tau tangles and lipid accumulations,” Cohen said.
“The first two got a lot of attention. The next frontier is lipids. Since APOE is the largest genetic risk factor, we believe it holds the clues to how lipids fit into the story.”
The role of lipids in the development of Alzheimer's disease
Neurons in the brain coexist with and rely on many other cell types to function properly. Astrocytes, which take their name from their star shape, ensure the survival of neurons by nourishing and detoxifying them with the help of a multifunctional protein, APOE.
One of the three forms of this protein, APOE4, significantly increases the risk of developing Alzheimer's disease, but the mechanisms at play are unknown.
A collaboration between the University of Geneva (UNIGE), the European Molecular Biology Laboratory (EMBL), the University of Zurich and the pharmaceutical company AbbVie has discovered a potential mechanism: far from stopping working, APOE4 is instead more efficient.
By activating astrocytic lipid secretion, it causes the accumulation of potentially toxic lipids that are harmful to neurons and therefore could contribute to the development of Alzheimer's disease.
These findings, published in the journal Cell Reports, shed new light on the neurodegenerative mechanisms of a disease that affects nearly 50 million people worldwide.
Astrocytes, present in large numbers in the brain, have an important protective function. These cells secrete apolipoprotein E (or APOE), a small protein that forms particles containing lipids and vitamins to nourish neurons.
It also detoxifies neurons by freeing them from “lipid waste” which could become harmful if not removed. Since neurons are unable to eliminate this waste on their own, APOE comes into play to collect it and bring it back to astrocytes where it is destroyed.
The gene that codes for APOE exists in three frequent variants in humans: APOE2, present in 8% of the population, APOE3, the most common, and APOE4, which is found in almost 15% of people and increases the risk to develop Alzheimer's disease by a factor of ten.
“The reasons why APOE4 increases the risk of Alzheimer's disease so significantly are not well understood,” explains Anne-Claude Gavin, professor at the Department of Cellular Physiology and Metabolism at the UNIGE Faculty of Medicine and holder of a chair from the Louis-Jeantet Foundation. , who led this research together with Viktor Lakics, a researcher and biological area leader in neuroscience discovery at AbbVie.
What are the mechanisms behind APOE4 dysfunction? And above all, could they serve as a basis for prevention or therapy? To answer these questions, Anne-Claude Gavin and her team joined forces with scientists from the European Molecular Biology Laboratory (EMBL), the University of Zurich and AbbVie.
Working on these questions, the research team identified new molecular mechanisms that explain how APOE binds to astrocyte membranes to sense and extract the lipids it needs.
Using human cell lines with different APOE variants, in vitro experiments have shown that APOE is very efficient at transporting potentially harmful lipids produced in neurons.
“And to our great surprise, the APOE4 variant turned out to be even more efficient than the other forms,” reveals Katharina Beckenbauer, former post-doc in Anne-Claude Gavin's group, senior scientist at AbbVie and one of the first authors of the study's work .
“So, contrary to what we thought until now, the problem is not that APOE4 stops working, but quite the opposite. And the mechanism goes haywire.”
As astrocytes age, they become less efficient and begin to accumulate lipids instead of destroying them.
“We modeled this process experimentally and observed the molecules secreted by astrocytes,” explains Karina Lindner, a Ph.D. student in Anne-Claude Gavin's laboratory and one of the first authors of this work.
“We observed that cellular aging diverts APOE from its primary function – transporting lipids to neurons and also recovering lipids from them – towards the secretion of triglycerides, particular lipid species that could become harmful if not removed.”
And this phenomenon is exacerbated by APOE4: it stimulates the secretion of triglycerides, leading to their uncontrolled accumulation.
This deleterious accumulation of potentially harmful lipids may well be a major contributor to neuronal death, a hallmark of Alzheimer's disease. “APOE4 would therefore have the ability to accelerate the pathological process of the disease through the mechanism we have discovered.”
To better understand the details of the action of APOE and especially of the E4 variant, UNIGE scientists now want to determine how the secretion of these potentially harmful lipids is regulated and whether this secretion can be detected in people suffering from Alzheimer's disease.
#Alzheimer39s #probable #main #accumulation #fat #brain #cells






