By studying the ANK2 gene, the scientists ofUniversity of North Carolina at the Chapel Hill School of Medicine and colleagues showed that its rare variants, constantly present in individuals with autism spectrum disorder (ASD), can alter the architecture and organization of neurons, potentially contributing to autism and neurodevelopmental comorbidities.
The results of the discovery have been published in the scientific journal eLife.
Gene ANK2: Here’s what the research says
The discovery was led by Damaris Lorenzo, Ph.D., assistant professor at the UNC Department of Cell Biology and Physiology and member of the UNC Neuroscience Center and UNC Intellectual and Developmental Disabilities Research Center.

ANK2 instructs neurons and other cell types how to make anchirin-B, a protein with multiple functions in the nervous system. ANK2 encodes various versions (isoforms) of ankyrin-B through a process known as alternative splicing, whereby portions of the protein are excluded in the final molecules.
Mammals, such as mice and humans, express the full-size (giant) anchyrin-B isoform only in neurons; another very abundant isoform half its size is found in virtually every type of cell and organ. Several genetic studies have consistently identified rare variants in ANK2 in individuals with ASD, making it one of the high-risk genes associated with the condition.
Depending on the type and location in the gene, ANK2 variants can only affect giant anchor-B or both isoforms at the same time.
“Together with its high prevalence and striking clinical presentation, the uncertain cause of ASD is an important limiting step in the advancement of therapeutic options “, Lorenzo said. “The evidence of the genetic origin of ASD is strong but also complex, with at least 100 other high-risk genes linked to the disorder “.

Detecting the cause of ASD is made even more complicated because single genes such as ANK2 and the isoforms they encode can have more cellular functions. However, based on how subgroups of these genes functionally overlap or work together to activate biological pathways, scientists have proposed converging mechanisms that can be predominantly affected in individuals with ASD.
One such common mechanism is neuronal communication, which is driven in part by alterations in axons, the long extensions that carry signals from neurons to other neurons.
Underlying these processes within a single neuron is the axonal cytoskeleton, a complex network of filament-like proteins that play critical roles in the growth, shape and plasticity of each neuron. The axonal cytoskeleton is thought to be another important functional axis involved in ASD.(2)
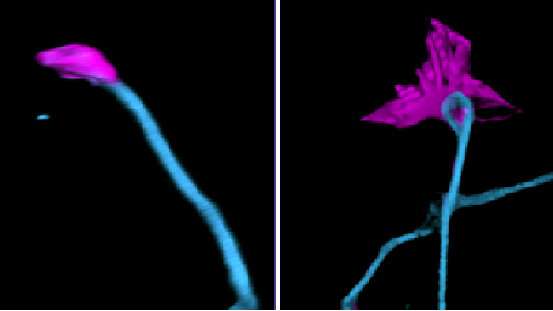
Lorenzo’s previous work published on JCB showed that the simultaneous loss of both major isoforms of ankyrin-B in the brain of mice caused profound anatomical defects involving axonal wiring, underscoring the importance of ankyrin-B in brain architecture and function.
In a later study published in PNAS , Lorenzo and colleagues at Duke University observed that deletion of only the giant anchyrin-B isoform in laboratory-grown neurons led to multiple axon branches, which implicated deficits in microtubule dynamics, an essential component of the cytoskeleton. .
In this new study, Lorenzo’s laboratory showed that selective loss of the giant anchyrin-B isoform leads to more axonal branches in the brain of mice and to volumetric increases in multiple axonal bundles including the corpus callosum. In collaboration with Eva Anton, Ph.D., at the UNC Neuroscience Center and co-author of the paper, the experts found that the giant isoform ankyrin-B is needed to maintain the topographical order of the calloused axons resulting from somatosensory cortex during brain development and to ensure specific targeting and refinement of callused projections on the opposite side of the brain.

The team did not observe increases in axon branches in a new mouse model they designed that lacks only the shorter ankyrin-B isoform: “These results confirm the diverging roles of the isoforms of anchor-B and support the critical and specialized roles of the giant anchor-B in the formation, targeting and refinement of collateral branches of axons “, said Lorenzo.
In collaboration with a team led by Meng Meng Fu, Ph.D., at the National Institute of Neurological Disorders and Stroke and co-authors of the article eLife , the researchers verified that the observed corpus callosum abnormalities did not involve changes in myelination and in the number and maturation of oligodendrocytes, a type of non-neuronal brain cells implicated in similar diseases.
“The cortical regions of the brain were the most directly linked to the pathology of ASD. The changes we observed in cortical structural connectivity likely stem from combined defects in axon branch initiation, guidance and pruning of erroneous or overabundant projections during development due to the gigantic anchyrin-B deficiency, “ Lorenzo specified.
Signals outside the cells modulate these processes to trigger changes in neurons through attractive and repulsive effects. Lorenzo’s research team showed that cortical neurons require the giant anchyrin-B isoform to make possible the repulsive effects of semaphorin 3A, a molecule that interacts with and collapses the tips of axons and their branches.
The team also showed that ANK2 variants that exclusively target giant ankyrin-B have a similar loss of response to the Semaphorin 3A molecule, revealing a possible mechanistic contribution to ASD.

“Our new insights along with our tools and methods will help us assign pathogenicity to other variants of ANK2. We are confident that there is an unknown biology relevant to brain function and ASD involving this gene and we are pursuing it “, said Lorenzo.
“Our bottom-up approach of functional discovery and validation contributes to the underdeveloped knowledge database of the functional etiology of ASD. This is fundamental because this heterogeneous and complex disorder probably requires personalized strategies for clinical intervention “, concluded the expert.
#ANK2 #gene #present #ASD #subjects #alters #neuronal #organization






